Initiative - Forming Fluorine Compounds from CFC Substitutes, or Greenhouse Gases
Category:News|Publishing : June 2, 2023
Hydrofluorocarbons (HFCs), one of the alternatives to chlorofluorocarbons, became widely used because they do not deplete the ozone layer; however, they were later found to be a greenhouse gas that causes global warming. Therefore, in the Kigali Amendment adopted in October 2016 at the 28th Conference of the Parties to the Montreal Protocol, it was decided to gradually reduce the use of HFCs by 2036.
Accordingly, in the future, it will be necessary for HFCs that have been manufactured to date and kept in storage in large volumes to be progressively incinerated as industrial waste. However, HFC incineration requires a large amount of energy and releases carbon dioxide at the same time, so its disposal has not progressed much. In recent years, there has been growing concern about illegal release of HFCs into the atmosphere. Therefore, the Shibata Laboratory in the Department of Engineering (Life Science and Applied Chemistry), Graduate School of Engineering, Nagoya Institute of Technology has started research and development of a method to convert HFCs to organic fluorine compounds that are essential to our lives.
In recent years, micro-flow synthesis, in which reagents and solvents are allowed to flow through a tubular channel to carry out reactions, has been the focus of attention. Micro-flow synthesis is a method that enables large-scale synthesis by continuously flowing a solution. If organic synthesis reactions can be applied to the flow synthesis method, it will be possible to make the method more closely resemble an industrial method. We have successfully developed a procedure of applying micro-flow synthesis to trifluoromethylation (CF3) using HFC-23 (HCF3: fluoroform) and to pentafluoroethylation (C2F5) using HFC-125 (HC2F5: pentafluoroethane). The procedure enables trifluoromethylation and pentafluoroethylation for various carbonyl compounds using the micro-flow equipment shown in the diagram.
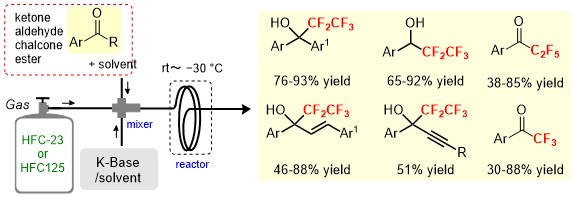
This procedure can be used not only as a method for recycling HFCs but also for instantly synthesizing high-value-added fluoroalkyl alcohols and fluoroalkyl ketones, which are expected to be used as pharmaceuticals and agrochemicals. Its further development is anticipated in various areas.
Our research findings are available in detail on our website, so feel free to take a look.
Vice-Chancellor of University of Tun Hussein Ong Malaysia (UTHM) visited NITech PRESS RELEASE - Successful terahertz wireless communication using a micro-resonator soliton comb: Expectations for next-generation mobile communications based on photonic technology

 Japanese
Japanese