RESEARCH NEWS - Mimicking the Earth's Crust: Examining Solidification of Mg-C-O-H Systems by Cold Sintering
Category:News|Publishing : September 20, 2022
Ceramic materials are ubiquitous in the world of construction. Building materials such as cement, bricks, tiles, or electrical insulators like porcelain are all ceramic products that we rely on in our daily lives. These ceramics are manufactured by a method called sintering ーthe process of turning powdery solids into a hardened mass by applying pressure or temperature. Most sintering processes involve temperatures beyond 1000 ֯ C, which makes this method very energy-hungry. Moreover, the high temperature also makes the sintering of raw materials such as carbonates and hydroxides difficult as they are prone to thermal decomposition at high temperatures.
Magnesium carbonate and hydroxides are emerging candidates for building materials owing to their thermodynamic stability and their ability to harden, or sinter, like slaked lime plaster. However, these materials cannot be sintered using the conventional sintering process as they undergo thermal decomposition. However, not much is known about how these materials react to a milder technique called cold sintering. To address this research gap, a team of researchers from Nagoya Institute of Technology, consisting of Prof. Shinobu Hashimoto and Mr. Keitaro Yamaguchi, investigated the mechanism by which Mg-C-O-H systems harden by the cold sintering process (or CSP). Their findings are summarized in a recent study made available online on April 21st, 2022, and published in Volume 48, Issue 15 of Ceramics International on August 1st, 2022.
The CSP gained popularity in recent years due to its low energy dependence. This process imitates the sedimentary rock formation process that occurs in the Earth's crust, allowing solidification to occur under several hundred megapascals of pressure but at milder temperatures such as 300◦C or below. This makes the process less energy-intensive and ideal for manufacturing construction materials with low thermal decomposition temperatures.
"Basic magnesium carbonate, or magnesite, has been proposed for use as a carbon storage material alongside its usage as a structural material. But magnesite is difficult to produce as by conventional industrial methods due to the influence of hydration during production and high temperature pyrolysis of sintering process," explains Prof. Hashimoto. "Our study aims to understand if Mg-C-O-H systems can undergo desirable solidification into construction ceramics via CSP."
The team used magnesium hydroxide and basic magnesium hydroxide powders as the ceramic precursors and water as the solvent. They heated the former at 250◦C and the latter at 150◦C with 10 mass% water, under a pressure of 270 megapascals (MPa) for an hour each. They found that compressive strength and relative density values for solidified magnesium hydroxide were 121 MPa and 84%, respectively, whereas the values for solidified basic magnesium carbonate were 275 MPa and 88%, respectively. The team also discovered that the water played a significant role in promoting the dissolution-precipitation reaction that is necessary for the densification of powders during the CSP. This phenomenon ensured that the sintering to form solid masses occurred at lower temperatures.
The results of this study provide a fresh perspective on sintering, which is generally considered a high-temperature and high-energy process. The CSP not only allows ceramics manufacturing of materials susceptible to thermal decomposition but also ensures excellent results by controlling the microstructure of the solidified products.
"The construction industry is one of the major consumers of energy responsible for 38% of global energy-related CO2 emissions. Through our research, we aim to get a step closer to building a future where manufacturing building materials is more sustainable and greener," concludes Prof. Hashimoto.
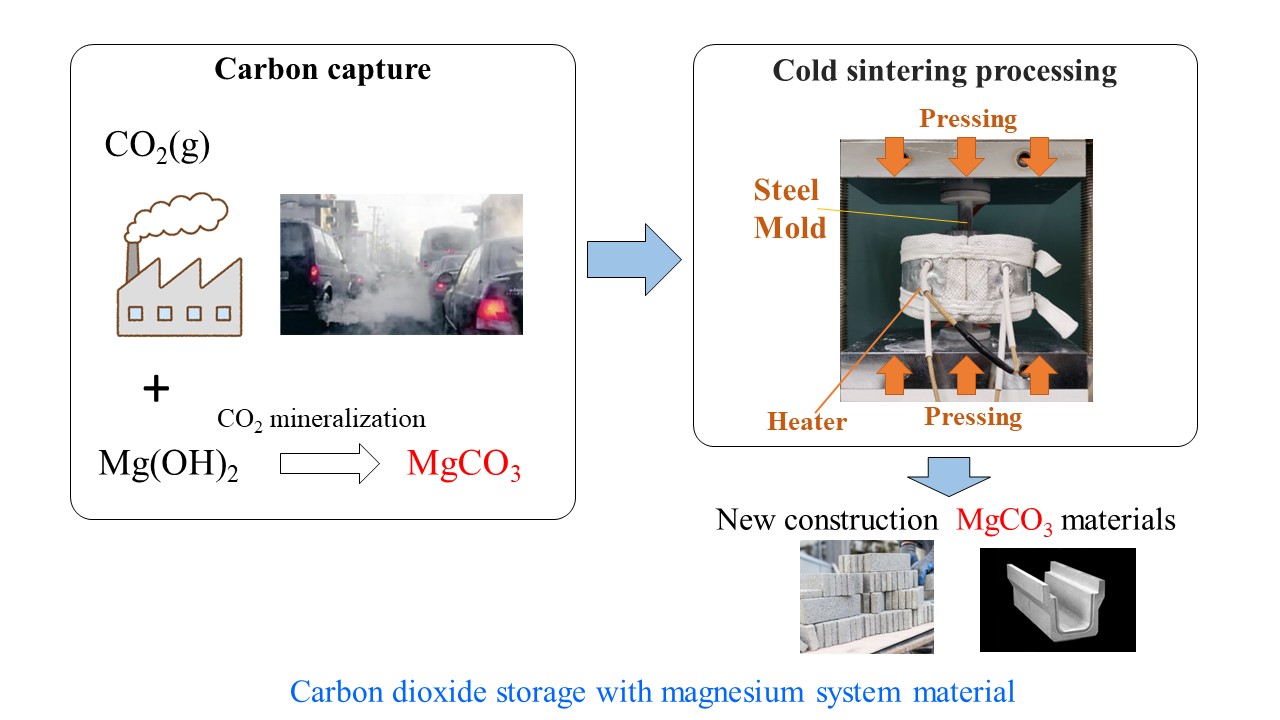
Applicability of the CSP to carbon capture and storage
The CSP emulates the sedimentary rock formation process in the Earth's crust and facilitates the solidification of basic magnesium carbonate and hydroxide powders. It may be used as a method to capture and store carbon dioxide and utilize them for the creation of construction materials
Image Credit: Shinobu Hashimoto from Nagoya Institute of Technology
Reference
|
Title of original paper |
Cold sintering of the Mg-C-O-H system |
|
Journal |
Ceramics International |
|
DOI |
|
|
Latest Article Publication Date |
August 1st, 2022 |
About Professor Shinobu Hashimoto from Nagoya Institute of Technology, Japan
Dr. Shinobu Hashimoto is a Professor and Research Faculty at the Department of Life Science and Applied Chemistry at NITech. He obtained his bachelor's, master's, and doctoral degrees from the Faculty of Engineering at Nagoya Institute of Technology. Prof. Hashimoto has authored over 140 publications with 1190 citations and an H-index of 17. His fields of expertise include inorganic, composite materials, and their surface and interface engineering. Prof. Hashimoto and his team are currently working on mineralogy, understanding crystal structure, and developing advanced ceramic fabrication methods.
Funding information
This study was supported by JST SPRING, Grant Number JPMJSP2112.
Contact
Professor Shinobu Hashimoto
TEL : +81 52 735 5291
E-mail : hashimoto.shinobu[at]nitech.ac.jp
Links : Details of a Researcher - HASHIMOTO Shinobu
*Please replace [at] with @ when contacting .
Open Campus 2022 held in August! RESEARCH NEWS - Preventing Dye Aggregation with Molten Salts to Improve Solar Cell Performance

 Japanese
Japanese